Clinical inflammation of the stroma of the lactating breast: NDC mechanobiological model
What is the NDC mechanobiological model of breast inflammation?
In 2001, Fetherstone proposed that mastitis results when intra-alveolar pressures rise so high that lactocyte tight junctions leak large milk proteins back into the stroma, triggering an inflammatory response.
Fetherstone was the first to question previous models of mastitis. Fethersone hypothesised that
-
Physiological responses to the breast during mastitis cause symptoms similar to those caused by pathogens
-
Lactocyte tight junctions leak proteins from the lumen into the tissues.1
In 2014 Ingman et al also pointed out that mastitis disease severity is associated with markers of inflammation rather than infection.2
Human milk and the mammary gland evolved from the innate immune system; lactation and inflammatory responses share many common mechanisms.3,4 I elaborate upon the NDC mechanobiological model of the regulation of milk production, here, to hypothesise that the dominant mechanism of clinical breast inflammation is mechanical hydrostatic pressure within the alveolus, which triggers microscopic and then clinical inflammation.
Building on new research about the mechanobiology of the lactating breast and the role of mechanosensing in the mammary gland immune response, a complex system perspective proposes that the mechanical effects of high intra-alveolar and intra-ductal pressure are a major regulator of the dynamic homeostasis of the lactating breast.
Elaborating on this model, breast stroma inflammation results in the following.5-13
-
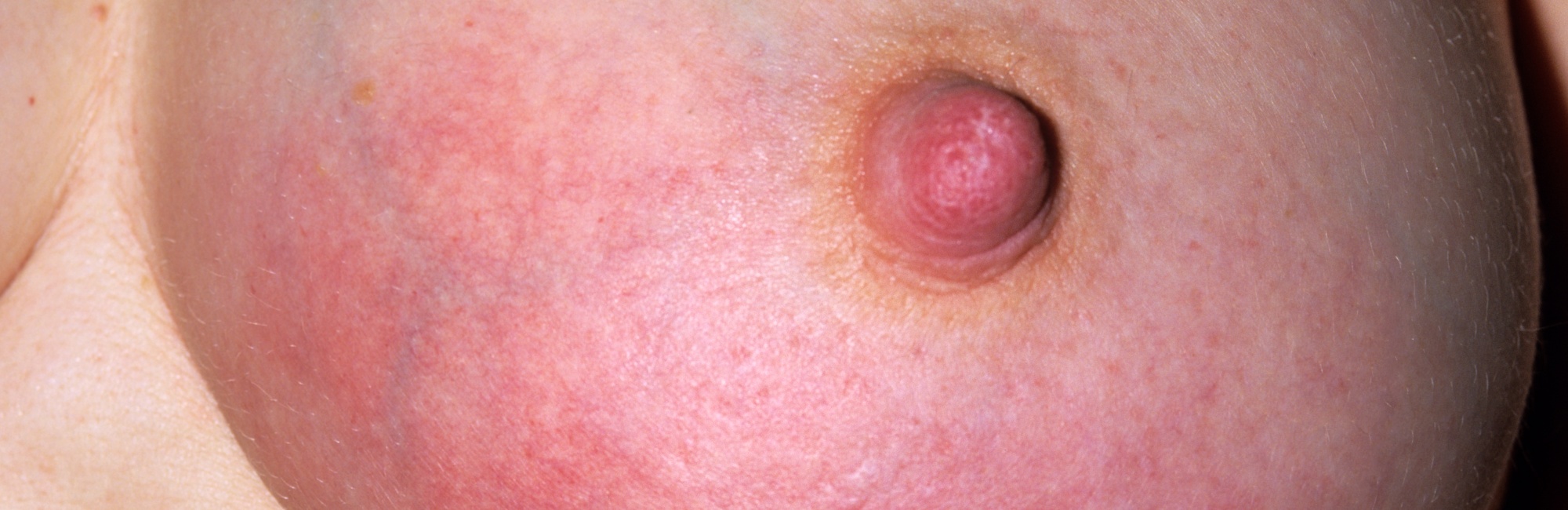
Once a critical mass of microscopic tight junction strain and alveolar rupture is reached within part of the breast, a clinically significant area of inflammation with hyperaemia, stromal tension, and perhaps tenderness or pain emerges.
-
If milk isn’t able to be extracted from a duct, for example, due to the compressive force of stromal tension or restrictive feeding practices, upstream ductal lumens and alveoli continue to dilate as lactocytes secrete more milk.
-
When inter-lactocyte tight junctions and alveolar basement membranes break, cell and molecular debris, leukocyctes, and interstitial fluid gather in the stroma.
-
Cellular and molecular waste and fluid pass into activated and dilated lymphatic capillaries.
-
A cascade of hyperaemia, increased interstitial fluid and lymphatic capillary dilation, increased stromal tension, increased ductal compression, increased intra-alveolar and intra-ductal pressure, and finally, alveoli rupture, ensues.
The mechanobiological model is consistent with Ingman et al’s 2014 hypothesis that partial involution occurs during lactation-related breast inflammation, resulting in decreased milk synthesis, which is observed post-mastitis.
What is the role of Toll Like Receptors?
Ingman et al 2014 proposed that inflammatory processes rather than pathogenic bacteria trigger breast inflammation in lactation. They observed that macrophages in the stroma surrounding the alveoli express Toll Like Receptors, as do lactocytes and mammary epithelial cells.2
But Toll Like Receptors are activated not only by bacterial and other stressors but by mechanical stress signals, initiating an inflammatory response. Toll Like Receptors are just one of multiple crosstalk mechanisms which detect and respond to endogenous cell and tissue damage, sensing and signaling within the complex adaptive system of the mammary gland immune system-milk interface.
What are the key mechanical factors which predispose to clinical breast inflammation?
Translating the mechanobiological model of breast inflammation into clinical practice, the following key mechanical factors elevate intra-alveolar and intra-ductal pressures and predispose to clinically relevant breast inflammation.5
-
Any factor which causes external compression of lactiferous ducts (for example, conflicting intra-oral vectors of force during suckling, which compress ducts);
-
Any factor which increases internal stromal tension and occludes lactiferous ducts (for example, micro-vascular trauma in the stroma resulting from lump massage or vibration);
-
Any factor which decreases frequency of alveolar contraction and ductal dilations (for example, spacing out feeds or of milk removal opportunities).
.jpg)
Recommended resources
Mechanical pressures are the engine room of breastfeeding and lactation
The NDC mechanobiological model explains downregulation of breast milk production
Adipocytes and mechanical sensing
Your breasts and milk evolved from an ancestral immune system
Selected references
-
Fetherstone C. Mastitis in lactating women: physiology or pathology? Breastfeeding Review. 2001;9:5-12.
-
Ingman WV, Glynn DJ, Hutchinson MR. Inflammatory mediators in mastitis and lactation insufficiency. Journal of Mammary Gland Biology and Neoplasia. 2014;19:161-167.
-
McClellan HL, Miller SJ, Hartmann PE. Evolution of lactation: nutrition v. protection with special reference to five mammalian species. Nutrition Research Reviews 2008;21:97-116.
-
Oftedal O. The evolution of milk secretion and its ancient origins. Animal 2012;6(3):355-368.
-
Douglas P. Re-thinking benign inflammation of the lactating breast: a mechanobiological model. Women's Health. 2022;18:17455065221075907.
-
Basree M, Shinde N, Koivisto C. Abrupt involution induces inflammation, estrogenic signaling, and hyperplasia linking lack of breastfeeding with increased risk of breast cancer. Breast Cancer Research. 2019;21(80):https://doi.org/10.1186/s31058-019-1163-7
-
Jindal S, Narasimhan J, Vorges VF, Schedin P. Characterization of weaning-induced breast involution in women: implications for young women's breast cancer. Breast Cancer. 2020;6(55):https://doi.org/10.1038/s41523-020-00196-3.
-
Kim T-J. Mechanobiology: a new frontier in biology. Biology. 2021;10(570):https://doi.org/10.3390/biology10070570.
-
Kobayashi K, Han L, Lu S-N, Ninomiya K, Isobe N, Nishimura T. Effects of hydrostatic ompression on milk production-related signaling pathways in mouse mammary epithelial cells. Experimental Cell Research. 2023;432:113762.
-
Noam Zuela-Sopilniak, Lammerding J. Can’t handle the stress? Mechanobiology and disease. Trends in Molecular Medicine. 2022;28(9):710-725.
-
Stewart TA, Hughes K, Stevenson AJ, Marino N, Ju AL, Morehead M, et al. Mammary mechanobiology - investigating roles for mechanically activated ion channels in lactation and involution. Journal of Cell Science. 2021;134:doi:10.124/jcs.248849.
-
Weaver SR, Hernandez LL. Autocrine-paracrine regulation of the mammary gland. Journal of Dairy Science. 2016;99:842-53.
-
Zaragoza R, Garcia-Trevijano ER, Lluch A, Ribas G, Vina JR. Involvement of different networks in the mammary gland involution after the pregnancy/lactation cycle: implications in breast cancer. International Union of Biochemistry and Molecular Biology. 2015;67(4):227-38.