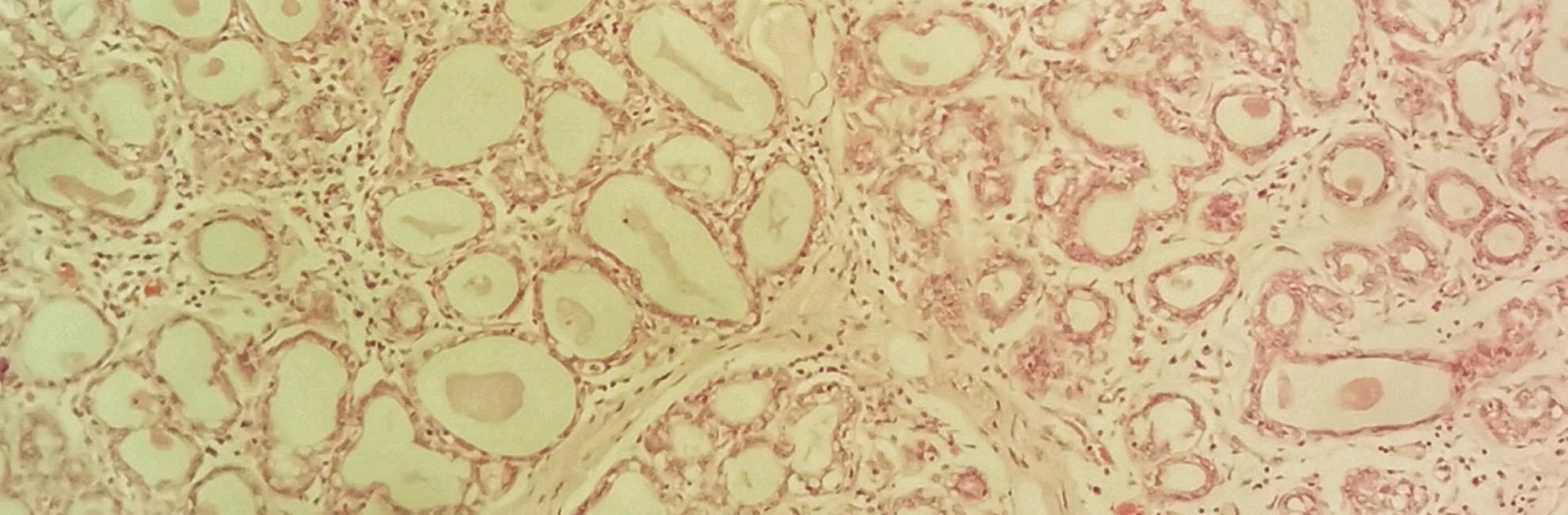
Research corroborates the NDC mechanobiological model of downregulation of breast milk production
Jindal et al 2020
The authors noted that three-dimensional time lapse imaging of the mammary gland of lactating mice supports the existence of a multifaceted system of mechanical sensing through chemical signals in the mammary gland.
For this study, breast tissue samples were obtained from a prospective non-intervention clinical trial in healthy women. Biopsies were obtained from the right quadrant of the right breast in 112 healthy women at nulliparity, lactation, and multiple postweaning time points, and analysed using quantitative immunohistochemistry.
The authors found that mammary remodeling programs observed in rodents are mirrored in the human breast.
There is a natural variation in lobule types, even within lactation. Lobules are categorised according to alveoli numbers: type 1 has 1-15 alveoli; type 2 has 16-50; type 3 > 50 alveoli per lobule; type 4 are lobules with a secretory morphology. Some lactating women had only 20% or less of type 4 lobules, others have 100% type 4 lobules. 20% of lactating women had < 60% of combined type 3 and 4 lobules at time of biopsy. During lactation, even small type 1 and 2 lobules express adipophilin, a protein essential for milk lipid secretion. That is, all lobular subtypes are secreting milk, whether 1-4. The authors hypothesise that partial gland involution occurs prior to the complete cessation of breastfeeding in response to decreasing milk removal.
Lobular composition of the breast was reduced by a fortnight post-wean and returned to pre-pregnant-like state by 3 months post-wean.
Weaning is associated with the stromal hallmarks of wound healing. Tight junction loss is a precursor to cell death of secretory epithelium. During the involution process, programmed cell death eliminates 80-90% of the secretory mammary epithelium within a tissue microenvironment which is skewed towards wound healing. That is, postweaning mammary glands have increased lympangiogenesis, fibroblast activation, fibrillar extracellular matrix deposition, and three-fold enrichment of macrophages and T-cells, compared to nulliparous glands. Epithelial cell death peaks at 2 weeks post-wean.
During lactation 50% of perilobular lymphatics are collapsed other 50 dilated. Lymphatics are implicated in clearance of milk and dying cells. The number of lymphatic vessels was highest in the one month postwean group, with new lymph vessel formation. At two weeks post-wean, all lymphatics were dilated, but the lymphatics were back to nulliparous levels by 3 months. Two weeks postwean, the authors found epithelial cell protrusion into the stormal comparment, showing that dying epithelial cells had escaped the confines of the alveolar structure and were entering lymphatic vessels.
The authors postulate that the biology of breast involution, not the biology of lactation, drives increased breast cancer risk in postpartum women.
Stewart et al 2021
Title: “Mammary mechanobiology – investigating roles for mechanically activated ion channels in lactation and involution”
Summary:
-
These authors describe the “force landscape” in the mature mammary gland and show that both luminal (milk-producing) and basal (milk-ejecting) epithelial cells experience mechanical forces during lactation.
-
They found increased expression of Piezo1, a mechanically activated ion channel, in the mammary epithelium during lactation. Luminal cells show functional expression of Piezo1.
-
However, deleting Piezo1 in luminal cells did not abolish lactation or involution, which suggests multiple mechanosensory pathways.

A closer look:
"Chemical and mechanical signals both play a role in [the function of the mammary gland]. However, despite this duality of input, much remains unknown about the nature and function of mechanical forces in this organ. Here, we characterize the force landscape in the functionally mature gland and the capacity of luminal and basal cells to experience and exert force."
The authors conducted three-dimensional time-lapse imaging of intact mammary tissue pieces from lactating mice.
When live myoepithelial (or basal) cells were visualised, a 32% decrease in their surface area was observed during myoepithelial cell contraction. Again using flourescent dyes, stochastic deformations to alveolar structures were noted at the same time.
Post-lactational involution has two stages in mice: the first is a reversible phase of 0-48 hours after cessation of infant feeding, after which lactation can still resume if milk removal continues. In this phase, milk accumulation in the lumen results in increased intraluminal pressure. This sustained overextension of the alveolar epithelium causes apical cell shedding, which prolongs epithelial barrier integrity by limiting cell density (that is, by causing stretching).
Luminal cell length and area were reduced by more than 50% by the end of the first phase of involution. The second irreversible phase from after 48 hours was marked by alveolar collapse, epithelial cell death and adipocyte regeneration.
Before an alveolus fills, lactocytes present rounded apices to the lumen. The authors visualised the same apical protrusions or curvature of the lactocyte which they found in mice alveoli in the alveoli of lactating human breast tissue.
When a lactocyte takes this columnar or triangular shape, fat droplets bud off from the apical cell membrane. As intra-alveolar pressure builds due to milk accumulation, lactocyte calcium-permeable ion channels are activated, and lactocytes absorb the increasing mechanical load by stretching and losing their apices. This protects inter-lactocyte tight junction integrity but prevents fat droplet extrusion. The lactocytes' apical membrane absorbs a substantial component of the pressure generated through milk stasis.
Kobayashi et al 2024
Title: “Effects of hydrostatic compression on milk production-related signaling”
Summary:
-
This study shows that accumulation of milk (i.e. hydrostatic compression in alveolar lumen) leads to downregulation of milk production in mammary epithelial cells).
-
The authors propose that mechanical pressure (from “too much milk”/“stasis”) can feed back to reduce secretion, consistent with a mechanobiological regulation of function.

A closer look:
Kobayashi et al's findings, working with in vitro mice lactocytes, that milk-production-related signaling pathways in lactocytes change in response to hydrostatic compression. Kobayashi et al conclude that hydrostatic ompression of the alveolar lumen may directly regulate milk production in the alveolar mammary epithelial cells, corroborating the NDC mechanobiological model which was published the previous year.
The authors investigated whether high hydrostatic ompression directly affects lactating mammary epithelial cells (MECs) or lactocytes from mice using a commercial compression device and a lactation culture model of MECs which have milk production ability and less permeable tight junctions. The mammary epithelial cells were cultured on cell culture inserts in differentiation medium containing prolactin and dexamethasons to induce milk component production and less permeable tight junction formation.
High hydrostatic ompression at 100 kPa for 8 hours decreased beta-casein and increased claudin-4 levels.Repeated rises and falls of the hourly hydrostatic compression induced activation of positive and negative signaling pathways for milk production, concurrently with stimulation of casein and lactoferrin production in MECs.
The results showed that high hydrostatic compression decreased milk protein production (caseins, lactoferrin), and altered both tight junction protein expression (weakened tight junctions) and milk production-related signaling molecules in lactocytes.
Two other relevant studies
Quaglino et al 2009 “Mechanical strain induces involution-associated events in mammary epithelial cells” (HC11 mammary epithelial cell line)
-
In cultured mouse mammary epithelial cells, application of radial stretching caused activation of signaling pathways like ERK1/2, increased expression of c-Fos, induced Leukemia Inhibitory Factor (LIF), STAT3 activation, and inhibition of AKT phosphorylation. These are events associated with involution (the process after weaning when milk production stops and tissue remodels).
-
This suggests mechanical stretching (from milk accumulation after suckling stops) can start the involution process.
Paavolainen et al 2021 “Integrin-mediated adhesion and mechanosensing in the mammary gland”
- Integrins and associated molecules (e.g., FAK, ILK) are involved in linking mammary epithelial cells to the extracellular matrix, transmitting mechanical cues. One of the findings is that integrin-linked kinase (ILK) in luminal epithelium mediates aspects of milk production in mouse models.
References
Jindal S, Narasimhan J, Vorges VF, Schedin P. Characterization of weaning-induced breast involution in women: implications for young women's breast cancer. Breast Cancer. 2020;6(55):https://doi.org/10.1038/s41523-41020-00196-41523.
Kobayashi K, Han L, Lu S-N, Ninomiya K, Isobe N, Nishimura T. Effects of hydrostatic ompression on milk production-related signaling pathways in mouse mammary epithelial cells. Experimental Cell Research. 2023;432:113762.
Paavolainen O, Peuhu E. Integrin-mediated adhesion and mechanosensing in the mammary gland. Seminars in Cell & Developmental Biology. 2021;114:113-125.
Quaglino A, Salierno M, Pellegrotti J. Mechanical strain induces involution-associated events in mammary epithelial cells. BMC Molecular and Cell Biology. 2009;10(55):https://doi.org/10.1186/1471-2121-1110-1155.
Stewart TA, Hughes K, Stevenson AJ, Marino N, Ju AL, Morehead M, et al. Mammary mechanobiology - investigating roles for mechanically activated ion channels in lactation and involution. Journal of Cell Science. 2021;134:doi:10.124/jcs.248849.